Correct answer : (3)
It can be solved as in the two following ways..
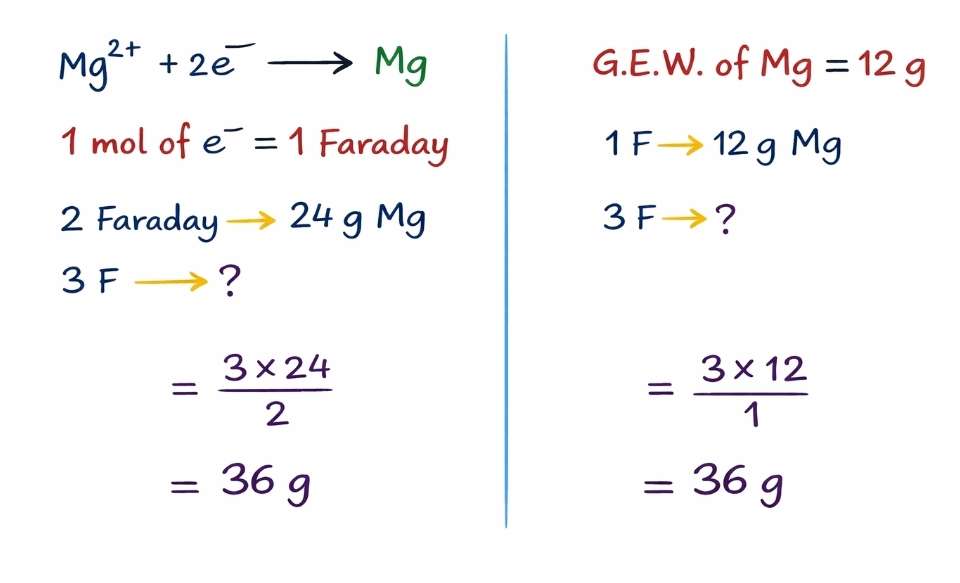 According to the of Faraday’s first law, the amount of substance deposited at an electrode or gas evolved or solute desolute is directly proportional to the quantity of electricity is passing through it.
According to the of Faraday’s first law, the amount of substance deposited at an electrode or gas evolved or solute desolute is directly proportional to the quantity of electricity is passing through it.
Passing of one Faraday current through an aqueous solution, Equivalent weight of a substance is deposited. When we pass one Faraday current through an aqueous solution of Magnesium bromide, 12 gm of Mg is deposited then how many grams of mg is produced after passing of three faraday of current which results three times of 12 will be deposited. That means, 36 grams of magnesium is deposited at the cathode when we pass 3F current through the solution.
There is an another way to calculate the amount of magnesium which is going to deposit after passing three Faraday of current. I, e.. one mole of magnesium is deposited when two faradays of electricity is passing through an aqueous magnesium Bromide solution. Then, the number of moles of magnesium is deposited after passing three faradays of current. It can be calculated as 3/2 = 1.5 moles of Mg.
Amount of the metal can be calculated by multiplying the atomic weight of metal by number of moles. 1.5×24=36gm is the amount of magnesium deposited at the cathode in the electrolysis.