 Answer : when Ethyl chloride reacts with KCN to form Ethyl cyanide, when which undergo hydrolysis to form propanoic acid.
Answer : when Ethyl chloride reacts with KCN to form Ethyl cyanide, when which undergo hydrolysis to form propanoic acid.
correct option: (1)
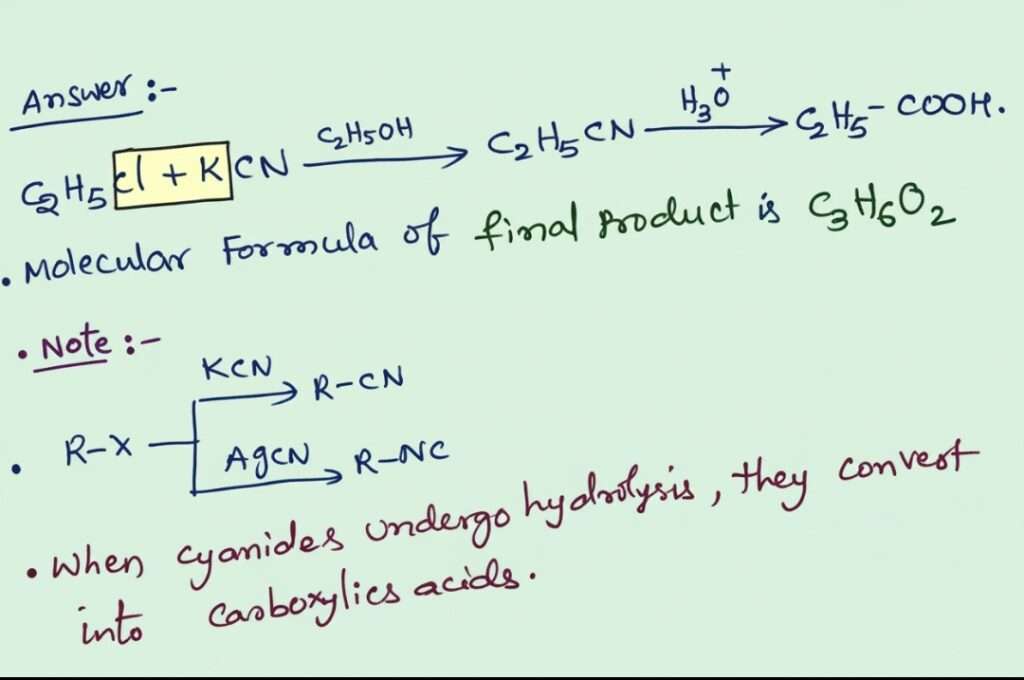
It is the conversion of alkyl halides into carboxylic acids through cyanide formation followed by hydrolysis. When Ethyl chloride reacts with potassium cyanide (KCN) in alcoholic medium, it undergoes SN2 reaction to form ethyl cyanide (propanenitrile).
In the reaction sequence, C₂H₅Cl + KCN ⟶ C₂H₅OH ⟶ H₃O⁺/Δ ⟶ Y. What is the molecular formula of Y? 1) C₃H₆O₂ 2)C₃H₅N 3)C₂H₄O₂ 4)C₂H₆O