Correct answer : (4)
Generally carbonyl compounds are reacting with HCN form Cyanohydrin. These reactions are nucleophilic addition reactions. For instance, Aldehydes react with HCN to form aldehyde Cyanohydrin. In the same way key don’ts also react with HCN to form ketone cyanohydrins.
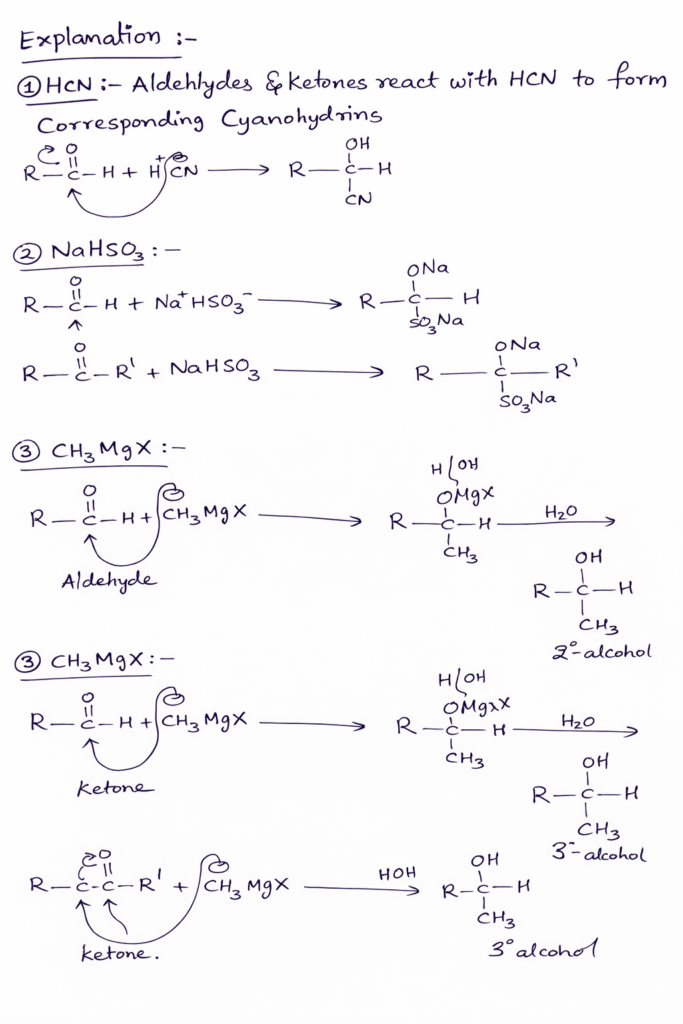
For example, Acetaldehyde reacts with HCN to form acetaldehyde cyanohydrin. Acetone or 2- propanone is reacting with HCN to form acetone cyanohydrin.
We need to understand one point, why is nucleophilic adition takes place rather than electrophilic addition. Oxygen is more electronegative than carbon in carbonyl group. Hence, The pi-bond polarises towards oxygen side and oxygen carries negative charge and carbonyl carbon gets the positive charge. Whenever nucleophile is attacking on the carbonyl carbon, oxygen carries the negative charge. If more electronegative atom carries the negative charge it should be stable.
If electrophile initially attacked the negatively charged oxygen atom, then the carbonyl carbon carries the positive charge it gets lower stable than the negatively charged oxygen. When there are two possibilities during the process of mechanism, major product will be obtained through the stable inter mediate. Hence, nucleophilic addition reactions are specifically taking place in these reactions rather than electrophilic addition reactions.