correct answer:(2)
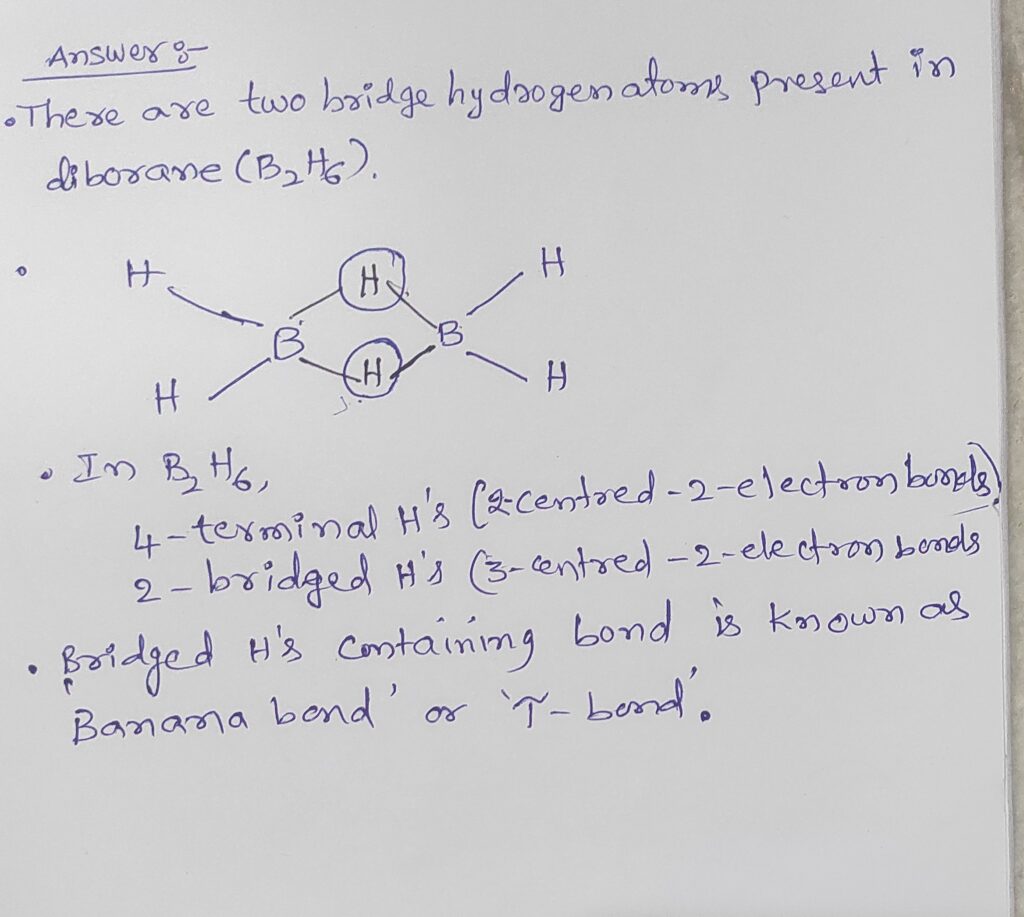
Structure of Diborane:
- The formula of diborane is B2H6.. Diborane consists of two boran Atoms connected by a bridge made of two bridges hydrogen atoms. Terminal Hydrogen Atoms: Each boran atom is bonded to two terminal hydrogen atoms and total of three hydrogen atoms around each boran.
- Bridged Hydrogen Atoms: The remaining to hydrogen atoms form a bridge bond between two boran atoms.
Video solution :
The number of bridge hydrogen atoms in diborane is 1)1 2) 2 3) 3 4) 4