Rate constant for the first order reaction:
The reaction in which the rate of reaction depends only on the concentration of one reactant is called first order reaction. The rate constant for the first order reaction can be determined in the following way,
A → Products
at t = 0 a (mol/lit) 0
after time t: a − x x
rate of reaction ∝ [reactants]ⁿ
Where, n = 1 (first order reaction).
− dx/dt ∝ (a − x)¹
− dx/dt = K₁ (a − x)
where
K₁ = first order rate constant
dx/(a − x) = − K₁ dt
On integrating the above equation,
∫ dx/(a − x) = − K₁ ∫ dt
− ln (a − x) = K₁ t + C …(1)
where,
C = Integration constant
When t = 0; then x = 0,
substitute in equation (1)
− ln (a − 0) = K₁ (0) + C
− ln a = 0 + C− ln a = C …(2)
Substitute (2) in (1),
− ln (a − x) = K₁ t− ln a
ln a − ln (a − x) = K₁ t
ln a/(a − x) = K₁ t
K₁ = (1/t) ln a/(a − x)
K₁ = (2.303/t) log a/(a − x)
∴ K₁ t = 2.303 log a/(a − x)
(i) Units:
K₁ = 2.303/t log a/(a − x)
K₁ = 2.303 /t log a/(a − x)
K₁ = 1/sec⁻¹
K₁ = sec⁻¹.
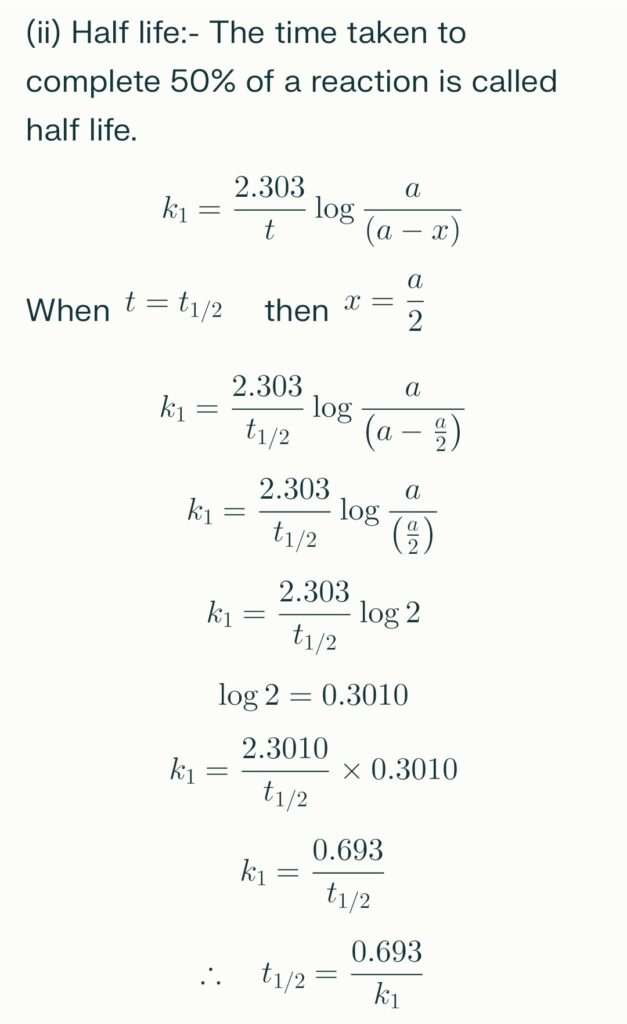 therefore the above formula explains that,
therefore the above formula explains that,
for a a first order reaction, half life is independent on the initial concentration of reactants.
It is the relation which is used to calculate the rate constant from half-life of first order reaction.
Examples of First-Order Reactions
decomposition of sulphuryl chloride to chlorine and sulphur dioxide.
SO2Cl2 → Cl2 + SO2 decomposition of nitrogen pentoxide to Nitrogen dioxide and oxygen.
2N2O5 → O2 + 4NO2 decomposition of hydrogen peroxide to water and oxygen
2H2O2 → 2H2O + O2