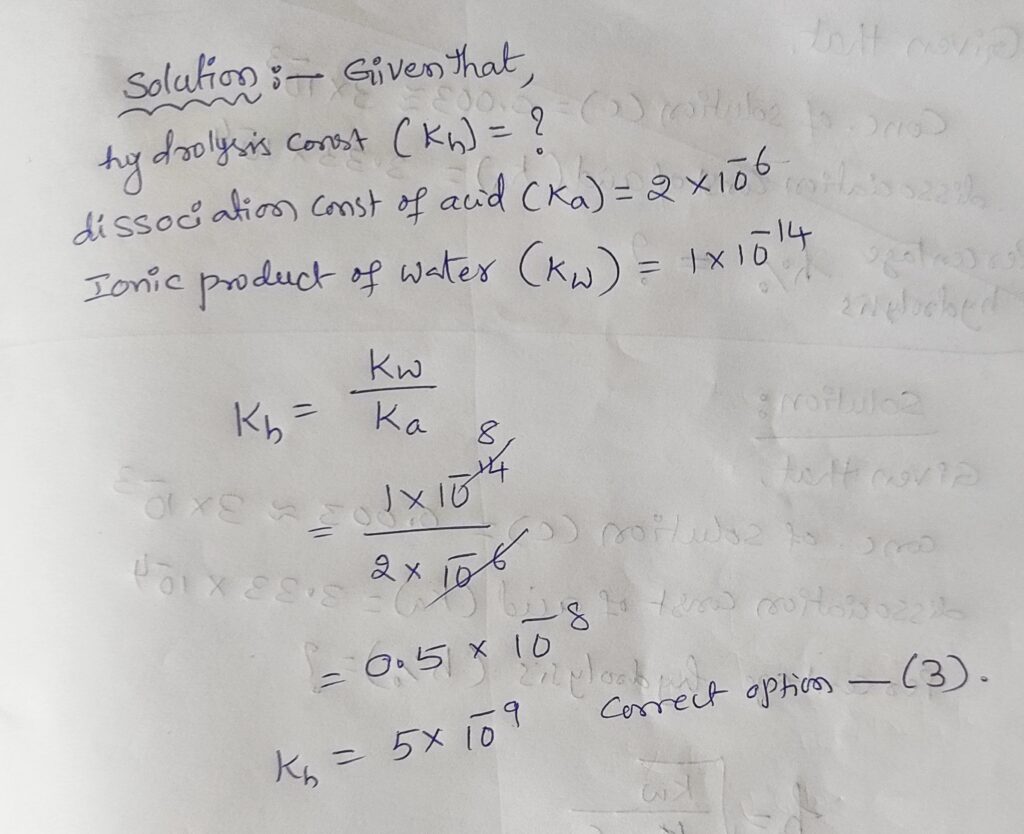
The hydrolysis reaction is a reversible reaction.The process of hydrolysis is a reverse reaction of neutralization.
hydrolysis constant (𝐾ℎ):The equilibrium constant for the hydrolysis reaction multiplied by concentration of water is known as hydrolysis constant. it is represented by Kh.
degree of hydrolysis (h): The fraction of the total salt which is hydrolysed it equilibrium is called degree of hydrolysis. It is denoted by “h”.
Calculate the pH of an aqueous solution of 1.0M ammonium formate assuming complete dissociation. ([ pK_a ] of formic acid = 3.8, [ pK_b ] of ammonia = 4.8)